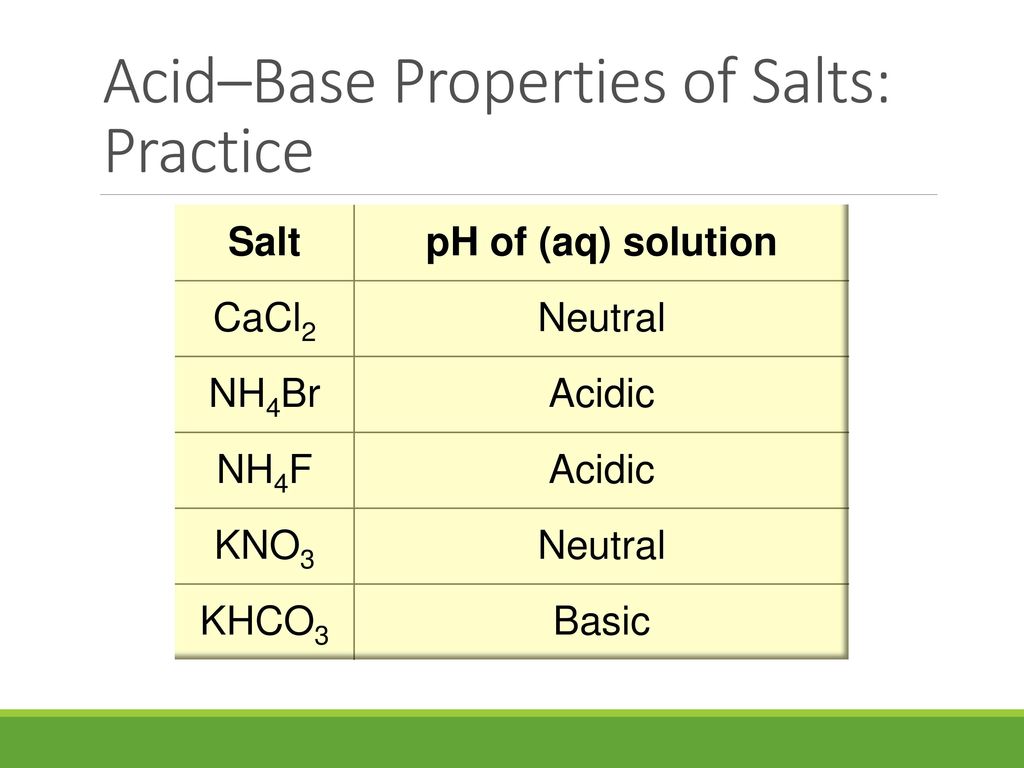
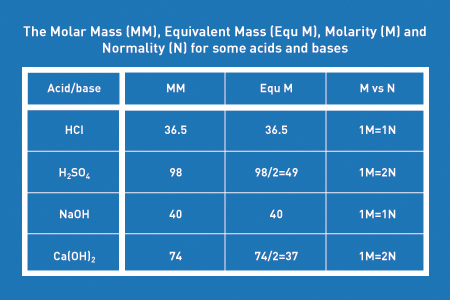
Name the acids and bases from which the following salts may be obtained. (i) Potassium sulphate (ii) Calcium chloride

Question Video: Calculating the Mass of Calcium Chloride That Contains a Given Mass of Chlorine | Nagwa

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

SOLVED: Classify the compounds as acids, bases, or salts. Which are Acids? Which are Bases? Which are Salts? Answer Bank LiCl KOH HBr Ba(OH)2 CaCl2 H2SO3 NH4NO3 H2C2O4

Pyrohydrolysis of CaCl2 Waste for the Recovery of HCl Acid upon the Synergistic Effects from MgCl2 and Silica | ACS Sustainable Chemistry & Engineering

![HCl Gas from conc. HCl(aq) and CaCl2 - [www.rhodium.ws] HCl Gas from conc. HCl(aq) and CaCl2 - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/equipment/pictures/calcium.hcl.gen.fig.gif)