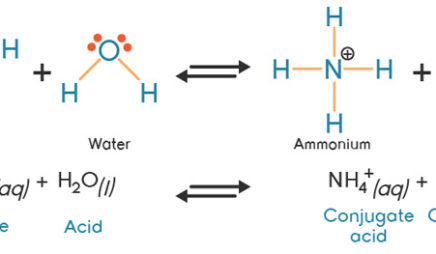
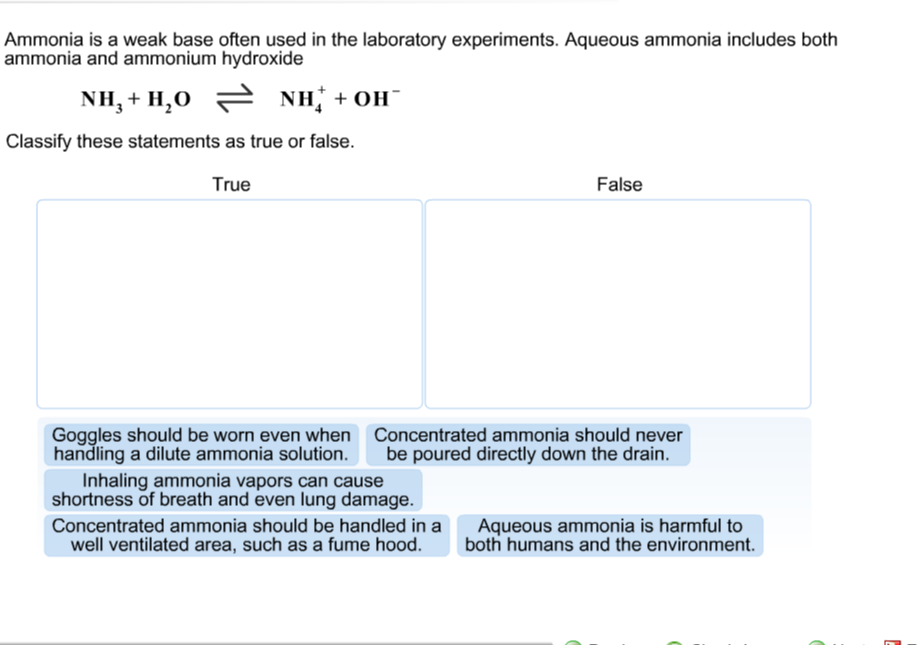
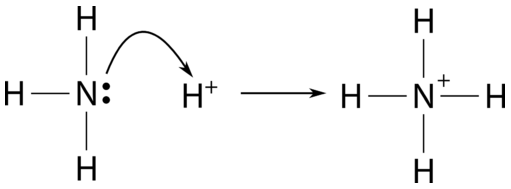
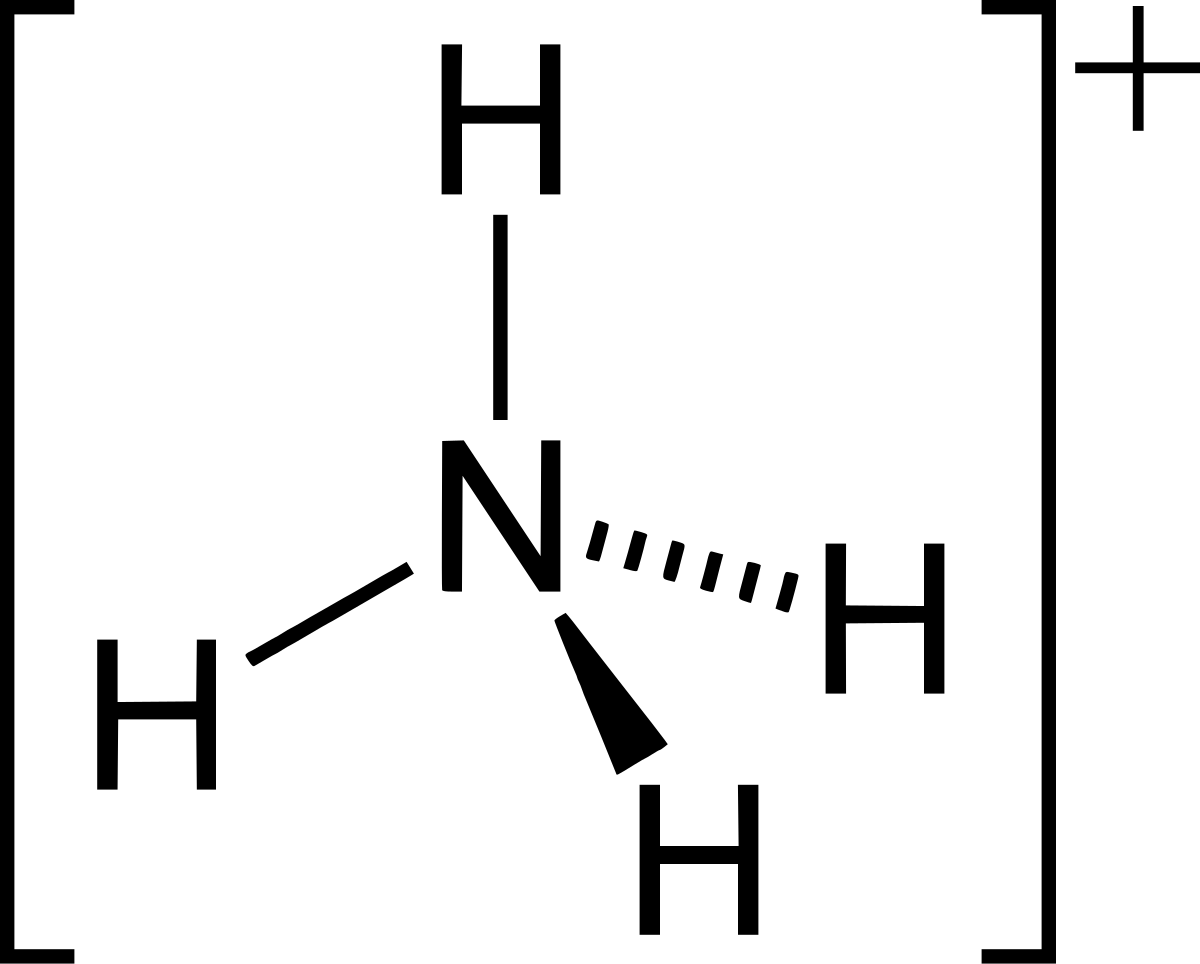
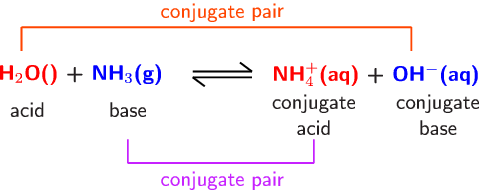
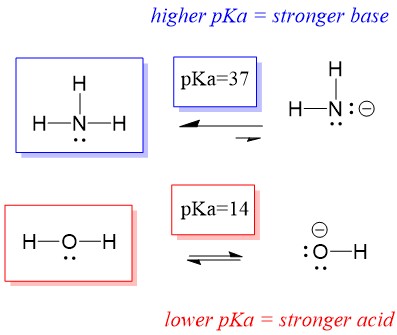
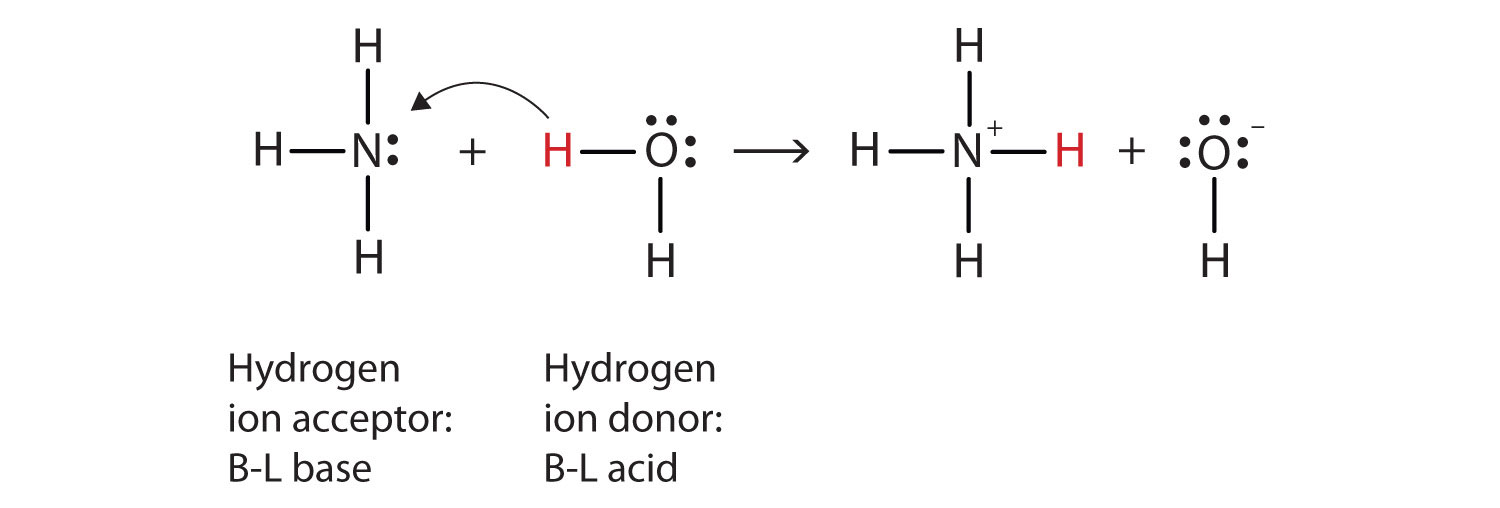
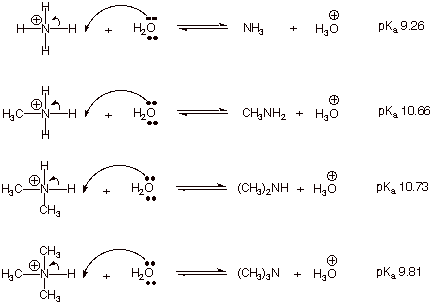A novel method for molecular transformation to obtain energy from ammonia! | Nature Portfolio Chemistry Community

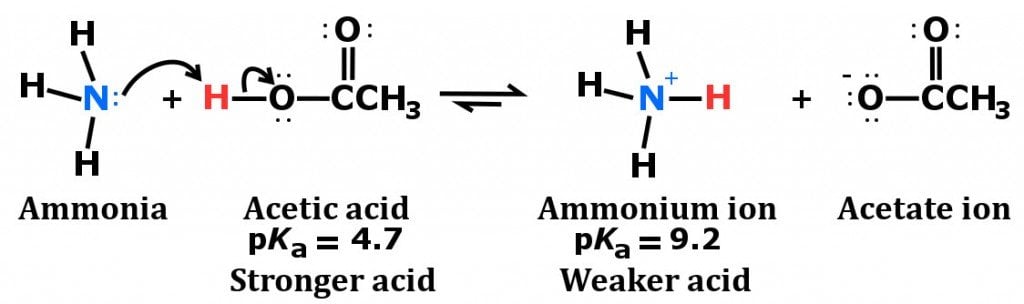
Spontaneity of the acid–base reaction between acetic acid and ammonia... | Download Scientific Diagram

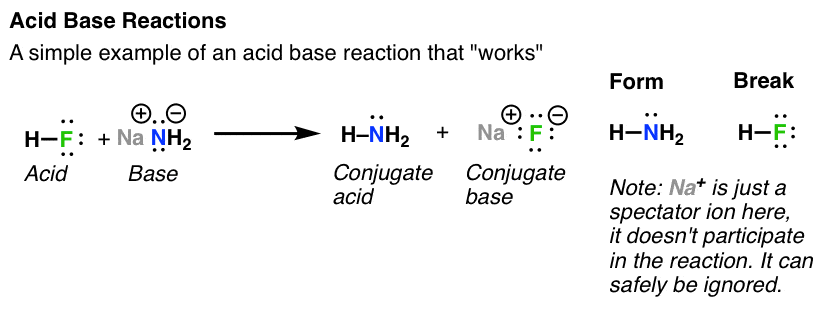
Question Video: Identifying the Lewis Acid in the Reaction of Ammonia with Boron Trifluoride | Nagwa